OcuRegen™
A topical bioelectric therapy for faster wound closure.
PCED beachhead. PD gated development. 24-week plan to lead nomination and in vivo closure signal.
OcuRegen™ is a defined eye‑drop program for impaired corneal epithelial repair. We start in PCED because endpoints are objective, time‑to‑signal is short, and local delivery is straightforward.
Program at a glance
Persistent epithelial defects are conventionally defined at ~2+ weeks of non‑healing. When closure fails, patients escalate through successive interventions; each step increasing cost, morbidity, and time‑to‑closure.
Target indication: Persistent Epithelial Corneal Defects (PCED)
Product: OcuRegen™ topical bioelectric modulators
OcuRegen™ focuses on candidates that preserve barrier function, raise corneal wound ion flux, and accelerate closure. By modulating ion channels (Na⁺/Cl⁻ flux) and maintaining barrier integrity, the wound’s electric field is restored and directional epithelial migration accelerates toward the injury.
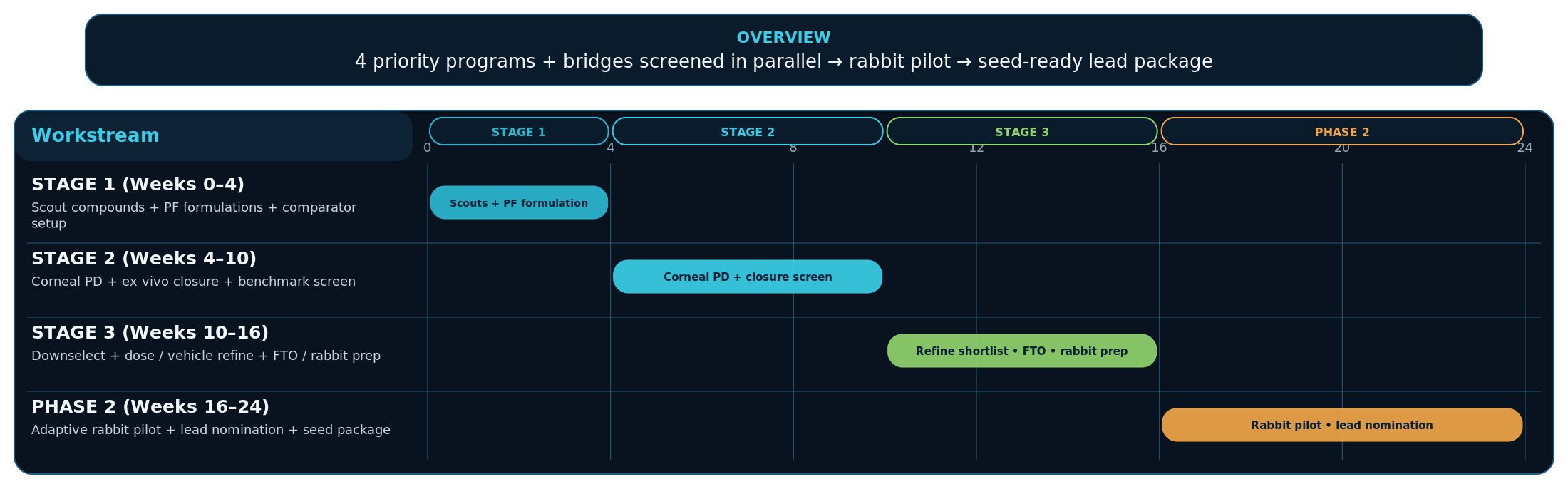
24-week plan: downselect → in vivo signal → seed‑ready package
16‑week PD‑gated downselect followed by an 8‑week non‑GLP rabbit pilot.
Outputs:
Week 16: PD‑validated ranked downselect + protocol locked
Week 24: rabbit efficacy signal + nominated lead + provisionals filed
PCED enables short trials with objective epithelial closure endpoints. We plan to pursue Orphan Drug + Fast Track while building mechanistic PD→closure linkage and preparing IND‑enabling work post‑seed.
Clinical & regulatory path
PCED is the beachhead, followed by expansion into neurotrophic keratitis (NK) and broader impaired-healing surface indications, leveraging the same approach.
Wedge & expansion
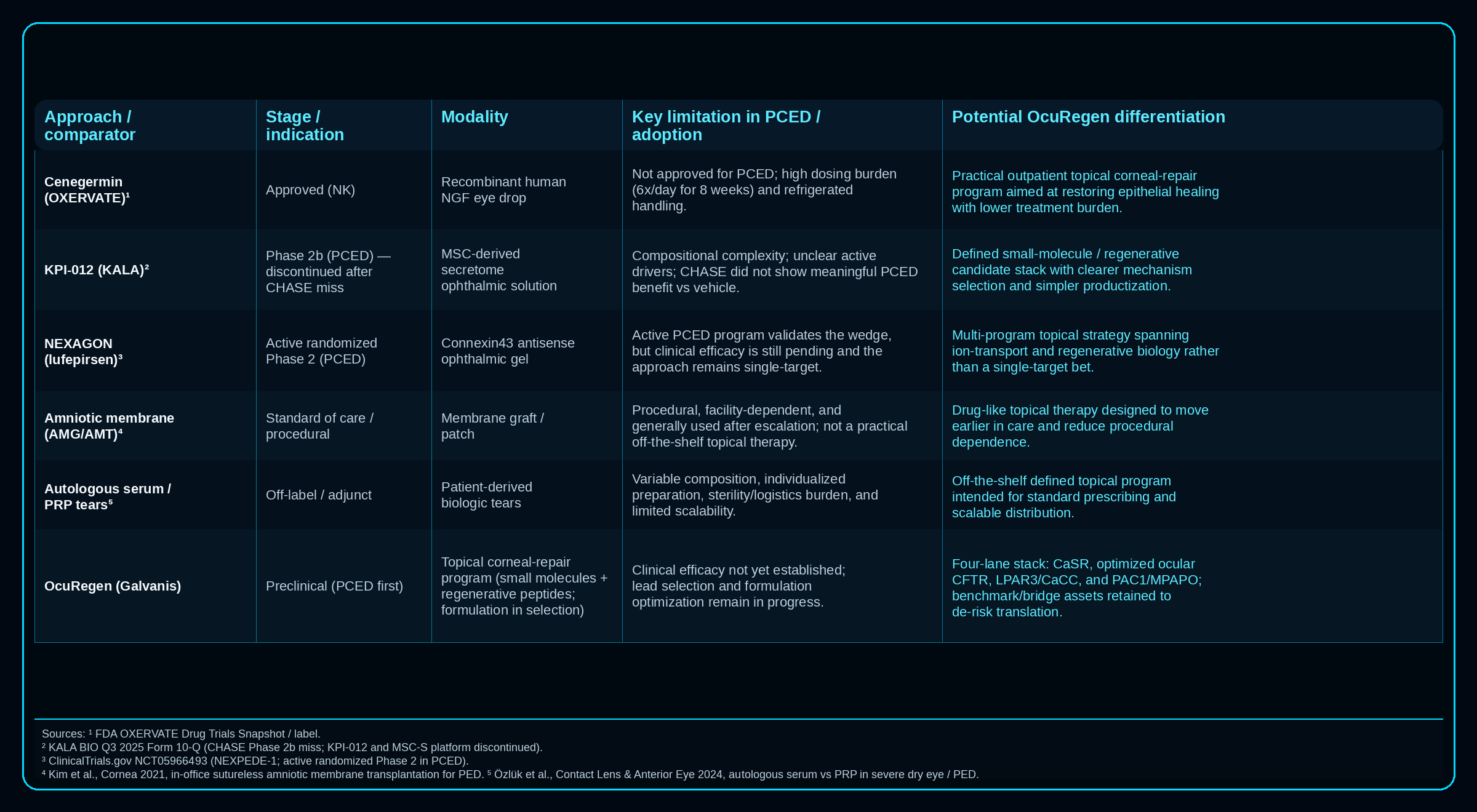
Differentiation
Moat/IP & risk controls
IP STRATEGY
File provisional patents on the peptide fusion composition of matter (CoM) and its use in corneal healing; provisional filings on 1) composition: peptide fusions/variants, 2) methods: modulating corneal bioelectric PD to accelerate epithelial closure, 3) assays/gating thresholds as know-how. Follow with PCT to cover EU/JP.
FREEDOM-TO-OPERATE (FTO)
Preliminary landscape scan complete; formal FTO search planned pre‑seed/early seed. We will review patents on similar peptides and device patents to ensure no overlap.
REGULATORY STRATEGY
Orphan Drug Designation precedent exists in PCED and we will apply to secure 7-year exclusivity. ODD + Fast Track are the near-term targets; breakthrough is pursued after clinical signal.
LOW POTENCY/ FAILURE IN VIVO
Early GO/NO-GO gates in ex vivo wound assays; expand lead library.
IMMUNOGENICITY
Sequence design to minimize T-cell epitopes; screen in silico & in vitro; confirm tolerability in preclinical tox.
REGULATORY/CMC DELAY
Engage ophthalmic-experienced CDMO; design formulation & analytical methods in parallel with efficacy.
IP CHALLENGES
File broad composition & method claims; build defensible know-how (formulation, assays, PD thresholds) as trade secrets.
OCULAR TOLERABILITY / CORNEAL TOXICITY
Corneal staining + IOP + histology + inflammation readouts.
OFF-TARGET ION TRANSPORT EFFECTS
Off-target ion transport effects (IOP, epithelial barrier).
Get the investor package
Request the deck and one pager or reach out directly to discuss diligence and collaboration.