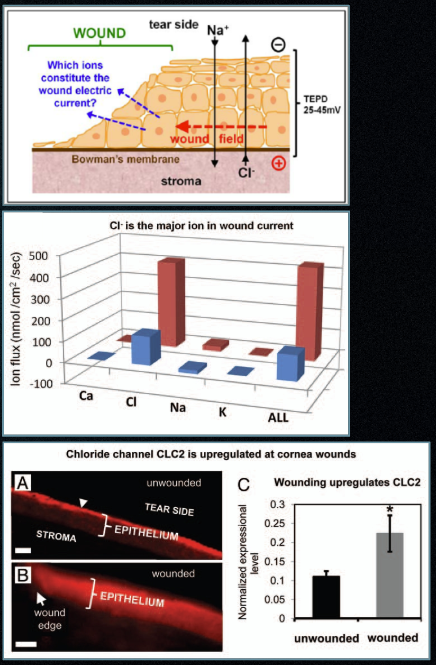
The corneal epithelium maintains a transepithelial potential through active ion transport and barrier integrity. When the epithelium is wounded, that potential partially collapses and creates endogenous electric fields oriented toward the defect. Corneal epithelial cells respond to physiologic electric fields with directional migration.
Human donor corneas show those wound currents can be pharmacologically increased. Chloride is a major carrier of corneal wound current, and both aminophylline and chloride-free conditions have been shown to increase wound current in human corneal tissue.
Galvanis is screening topical candidates that restore these signals while preserving epithelial barrier function.
WHAT WE MEASURE
Primary PD: ΔTEP / Δion flux
Primary function: scratch and ex vivo closure kinetics
Advancement rule: PD signal first, animal spend second
Mechanism-confirmed bioelectric healing
Endogenous electric fields (EFs)
Wound currents create lateral fields oriented into the defect.
Quantifiable pharmacodynamics (PD)
Measure ΔTEP / Δion flux and relate to closure kinetics.
GO/NO GO Gates
Advance only candidates meeting PD + closure thresholds.
Bioelectricity in corneal wound repair
The corneal epithelium maintains a transepithelial potential difference (TEP/TEPD) via active ion transport and tight junction resistance. When the epithelium is wounded, resistance drops and the TEP partially collapses (“short-circuit”), driving ionic currents toward the defect. Those currents generate lateral electric fields oriented into the wound, and corneal epithelial cells migrate directionally in physiologic electric fields.
Galvanis is building PD-gated topical bioelectric therapies for corneal repair. Corneal wounds generate measurable endogenous electric currents, and human donor corneas show those currents can be chemically modulated. Our pre-seed program does not bet the company on a single modality belief. Only candidates that preserve barrier function, increase corneal wound TEP, and improve closure advance. That gives OcuRegen™ a faster developmental path.
OcuRegen™: a topical bioelectric modulator
BIOELECTRIC MECHANISM
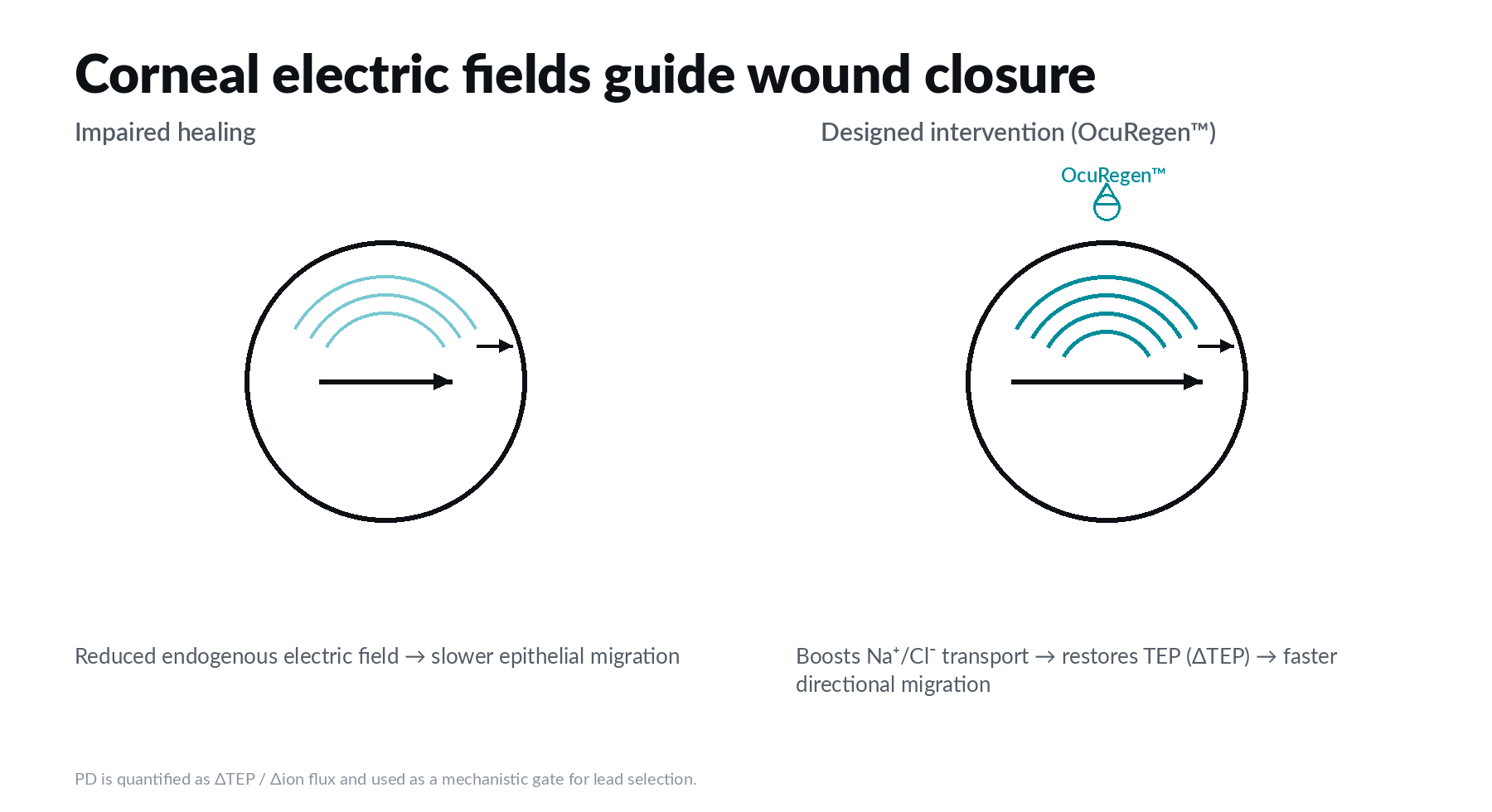
“Designed to boost Na⁺/Cl⁻ flux at the wound edge and restore the wound’s electric field.
PHARMACODYNAMIC GATED DE-RISKING
Advance only candidates meeting pre-specified PD thresholds (ΔTEP) plus functional closure improvement ex vivo.
ADDITIVE WITH CURRENT CARE
Designed to be used alongside existing therapies to further accelerate healing.
OPHTHAMOLOGY-READY
Preservative-free formulation goals with standard ocular safety/tolerability gating.
Restore the wound’s electric field to guide closure.
Corneal wounds generate endogenous electric fields that drive directional epithelial migration. In impaired healing, the electric field is reduced, slowing closure. OcuRegen™ is designed to modulate epithelial ion transport to restore transepithelial potential (TEP) and accelerate directional migration toward injury.
Mechanistic PD readout: ΔTEP / Δion flux.
Lead selection gated by PD plus faster scratch + ex vivo closure.
Goal: ≥50% faster wound closure vs. standard of care (preclinical benchmark target).
A name with lineage: Luigi Galvani
In the late 1700s, Italian physician Luigi Galvani observed that a frog’s leg could twitch in response to electrical stimulation; an early window into what he called “animal electricity.” His work helped launch electrophysiology and shaped how scientists think about bioelectric signals in living tissue. GALVANIS is a nod to that lineage: living tissues generate electric signals, and in modern biology we can measure and engineer those signals.